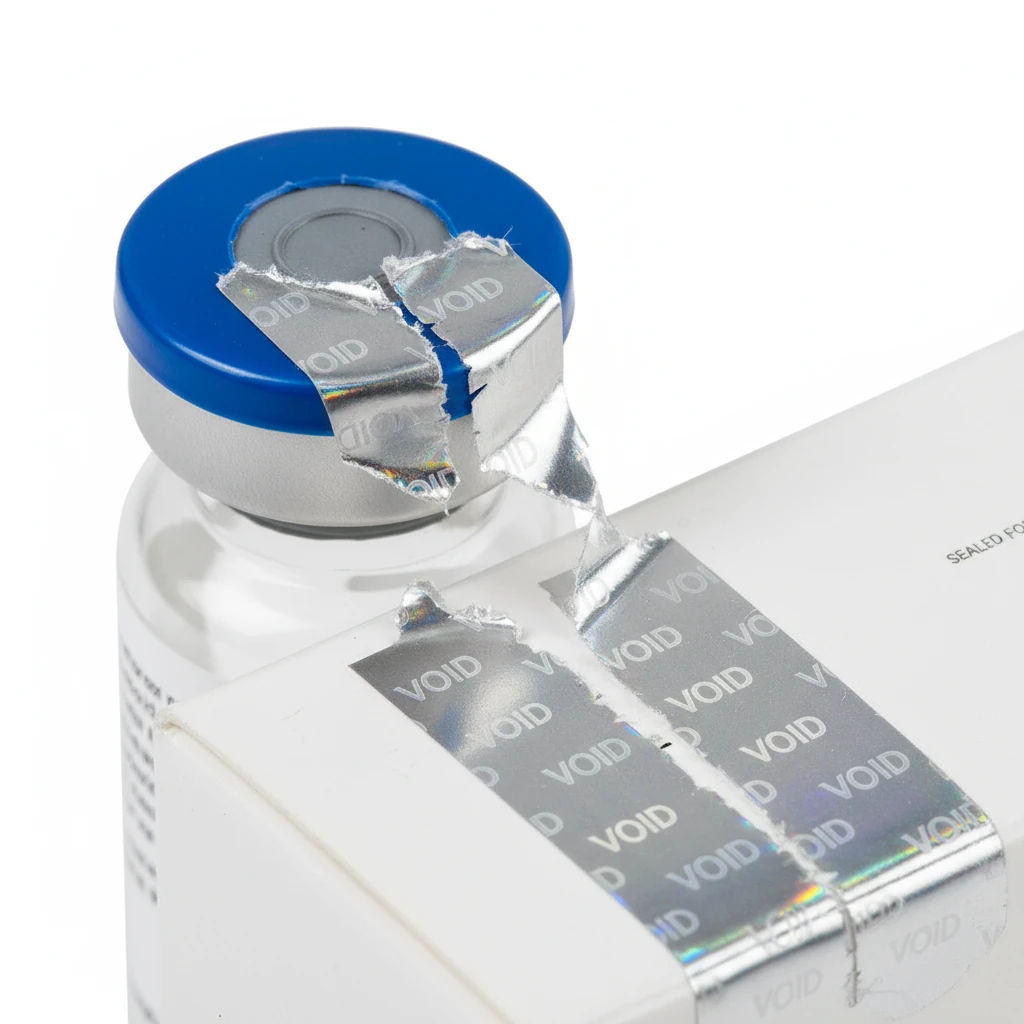
Pharmaceutical Labels
Compliant labeling solutions for pharmaceutical products with stringent quality and safety requirements
Pharmaceutical Labeling Excellence
PGPL Group specializes in pharmaceutical labeling solutions that meet FDA regulations and international standards. Our pharmaceutical labels ensure product integrity, patient safety, and regulatory compliance while maintaining the highest quality standards.
We understand the critical nature of pharmaceutical labeling and provide solutions that meet the most stringent requirements in the healthcare industry.
Designed for Regulated Healthcare Environments
From hospital injectables to OTC nutraceuticals
Acute & Chronic Therapies
Label layouts tuned for vials, blister packs and bottles used in critical care, chronic therapies and vaccines, with clear dose, route and storage information.
Global Regulatory Alignment
Workflows that reflect CDSCO, FDA 21 CFR and EU GMP expectations around legibility, data hierarchy, serialization zones and pharmacovigilance copy.
Controlled QC & Documentation
In‑house testing for adhesion, migration, freeze–thaw and print durability, backed by batch records and COAs that can plug into your QA documentation.
Pharmaceutical Label Solutions
FDA-compliant pharmaceutical labeling excellence with regulatory compliance and patient safety focus

Tablet Bottle Labels
Prescription bottle labels with clear dosage information, drug interactions, and patient safety warnings. Designed for optimal readability and compliance with healthcare regulations.
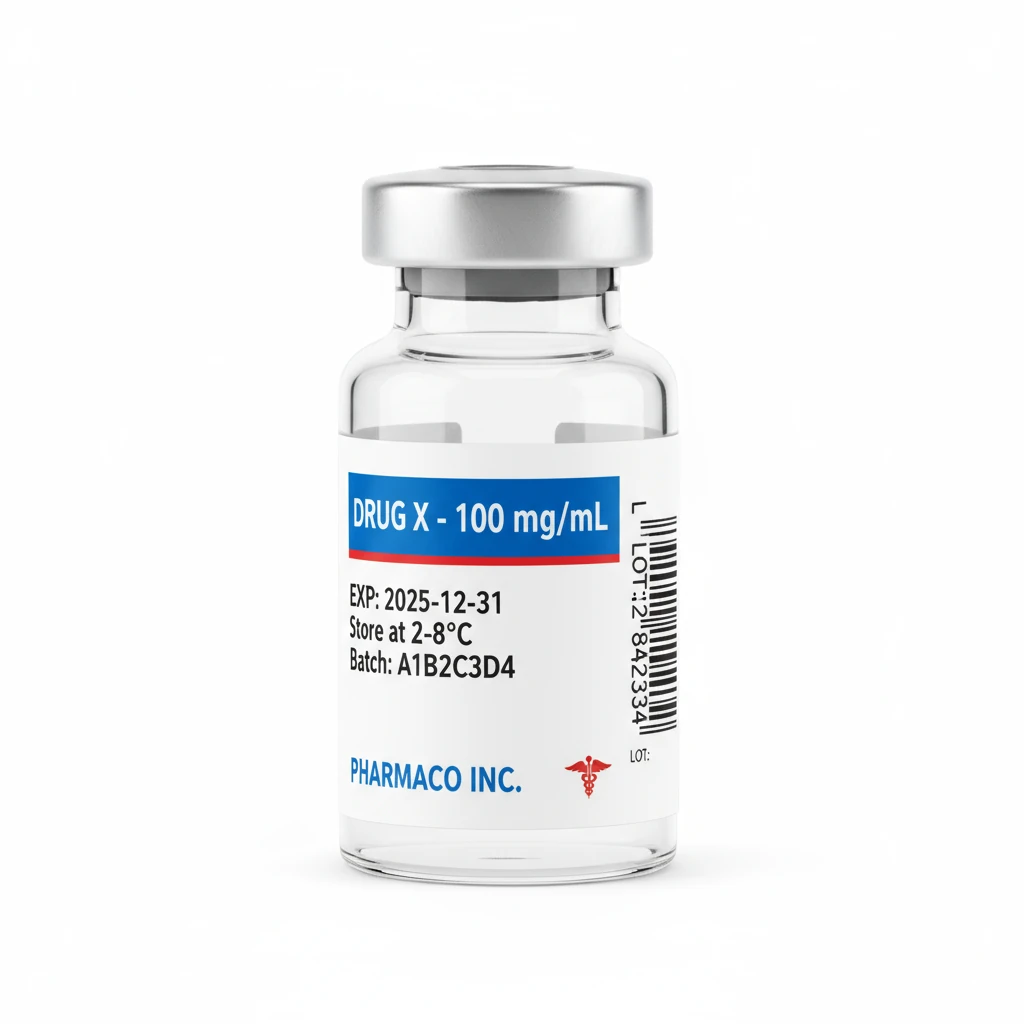
Injectable Vial Labels
Small format labels for vials and ampoules with critical information including concentration, expiry dates, and storage conditions for injectable medications.
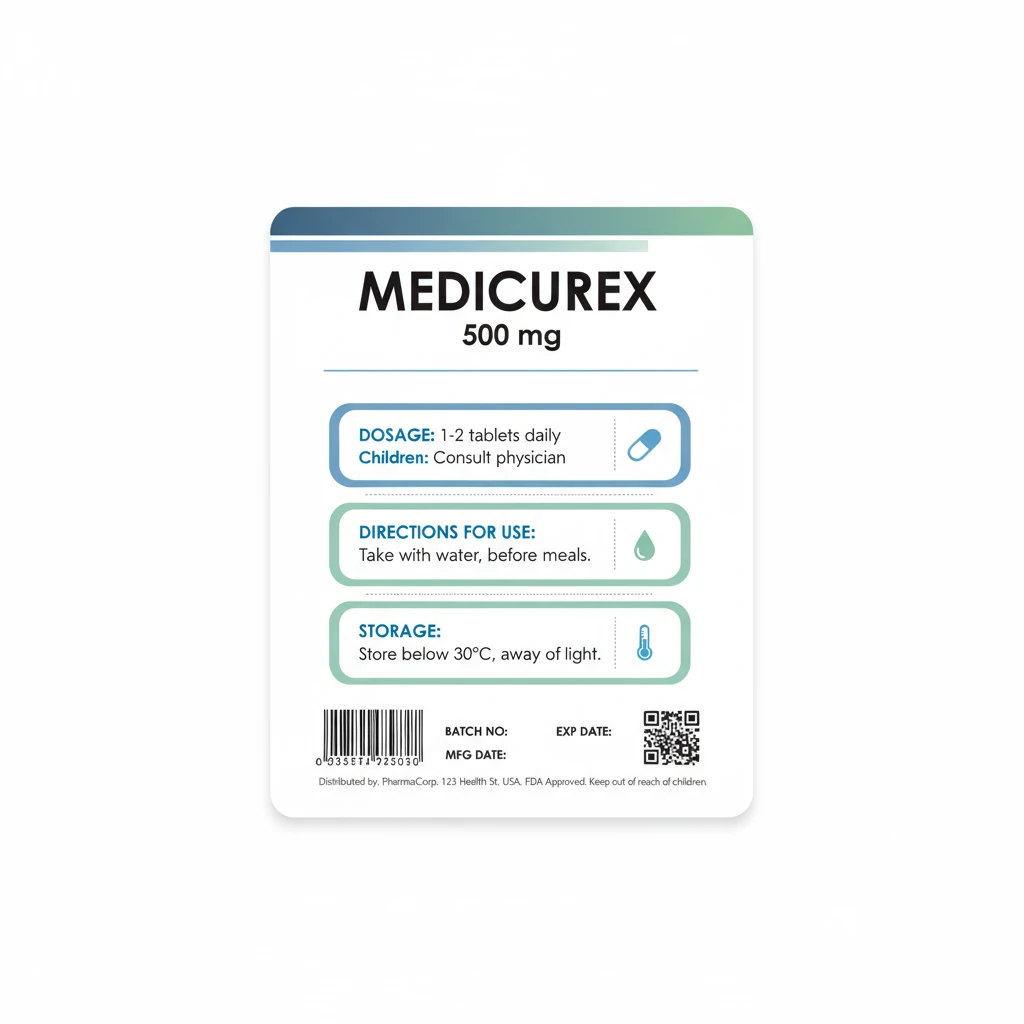
Carton Labels
Outer packaging labels for pharmaceutical boxes with comprehensive product information, barcodes, and regulatory compliance details for distribution and tracking.
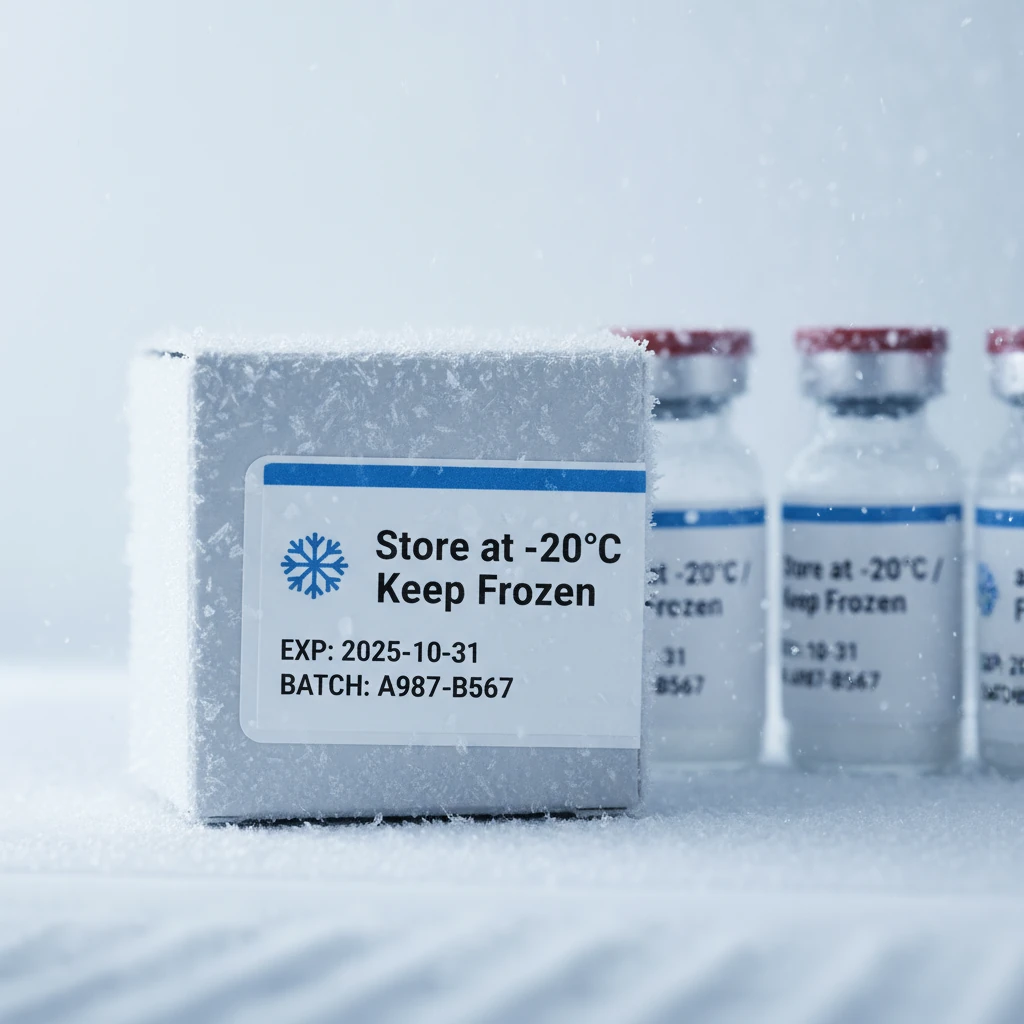
Cold Storage Labels
Freezer-grade labels for temperature-sensitive medications with special adhesives and materials that maintain adhesion and readability in extreme cold conditions.
Why Choose Our Pharmaceutical Labels?
Comprehensive solutions designed for healthcare compliance
FDA Compliant Materials
All materials meet FDA 21 CFR Part 211 requirements and are approved for pharmaceutical applications.
Tamper-Evident Security
Advanced tamper-evident features ensure product integrity and patient safety throughout the supply chain.
Cold Chain Resistant
Specialized materials and adhesives that maintain performance in refrigeration and freezing conditions.
Multi-Language Support
Support for multiple languages to meet global pharmaceutical labeling requirements.
Serialization Capability
Advanced serialization and track-and-trace capabilities for regulatory compliance and supply chain security.
Quality Testing
Comprehensive quality testing including migration testing, adhesion strength, and durability assessments.
Why PGPL TechPrint for Pharmaceutical Labels?
Print discipline built around QA, RA and production teams
Longstanding Healthcare Focus
For many years we’ve printed for pharma, biotech, nutraceuticals and hospital suppliers, so our teams speak the language of RA and QA.
Dedicated Pharma QC Cells
Special QC checkpoints for pharma work, including line clearance, sample retention and label–data verification before reels leave our floor.
Secure Artwork & Data Handling
Controlled access to artworks, serialization data and security plate sets helps protect sensitive product information.
Brands & Markets Served
We support prescription, OTC and export‑focused brands supplying to hospitals, pharmacies and modern retail across India and overseas.
Related Pharmaceutical Packaging Solutions
Combine primary labels with supporting security and information layers
Leaflet & Booklet Labels
Explore leaflet labels for IFUs and regulatory inserts when space on primary packs is not enough.
Security Features
Add holographic and tamper‑evident security labels to high‑risk or high‑value pharmaceutical SKUs.
Lamination & Protection
Use laminated constructions wherever cold‑chain, transport abrasion or cleaning chemicals could damage print.
Regulatory Compliance
Meeting global pharmaceutical standards
FDA Compliance
21 CFR Part 211 compliant materials and processes for US pharmaceutical market.
EU GMP Guidelines
Compliance with European Good Manufacturing Practice guidelines and standards.
ISO 15378
ISO 15378 certified materials for primary packaging of medicinal products.
Migration Testing
Comprehensive migration testing to ensure no harmful substances transfer to medications.
Ready to Ensure Pharmaceutical Compliance?
Contact our team today for a free consultation and quote for your pharmaceutical label requirements.